COVID-19 and its detection strategy in India
Hanjabam Rajanikanta Sharma *

COVID-19 testing in progress in Imphal on May 30 2020 :: Pix - TSE
CoVs are positive-stranded RNA viruses with a crown-like appearance under an electron microscope (coronam is the Latin term for crown) due to the presence of spike glycoproteins on the envelope.
COVID-19 pandemic is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). SARS-CoV-2 belongs to the betaCoVs category. It has round or elliptic and often pleomorphic form, and a diameter of approximately 60140 nm (NCBI).
The first case of COVID-19 in India, which originated from China, was reported on 30th January 2020. India currently has the largest number of confirmed cases in Asia, and has the third highest number of confirmed cases in the world after the United States and Brazil. A number of testing strategy is used in India since the first outbreak. Let us see in details how and where they are used.
1. Reverse TranscriptionPolymerase Chain Reaction (RT-PCR): Polymerase chain reaction (PCR) is a method by which a specific DNA sample is amplified into millions and billions of copies, thereby allowing scientists to study in detail from a very small sample of DNA.
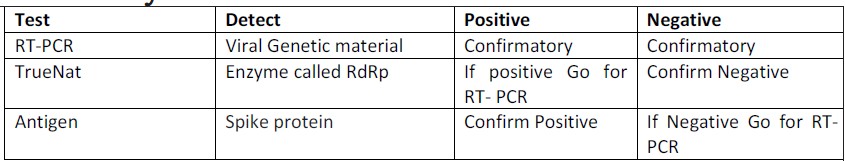
In the RT-PCR (Reverse transcriptionPolymerase chain reaction), the Viral Genetic material i.e. RNA is converted into complementary DNA (cDNA) before the amplification. RT-PCR uses RNA as starting material for in vitro nucleic acid amplification. The discovery of retroviral reverse transcriptase in the early 1970s ultimately made RT-PCR possible.
Reverse transcriptase is an RNA-dependent DNA polymerase, catalyzing DNA synthesis using RNA as the template. The end product is known as complementary DNA (cDNA). cDNA is not subject to RNase degradation, making it more stable than RNA. The cDNA serves later as a template for exponential amplification using PCR.
RT-PCR is commonly used in the diagnosis and quantification of RNA virus infections (e.g., human immunodeficiency virus and hepatitis C virus). Depending on the application, since the results of the PCR product can be determined, in real-time, it is also called real-time PCR or Quantitative polymerase chain reaction (Q-PCR).
RT-PCR and the COVID-19 virus
A sample is collected from the parts of the body where the COVID-19 virus gathers, such as a persons nose or throat. Then it is treated with several chemical solutions that remove substances such as proteins and fats and extract only the RNA present in the sample. Then the RNA is reverse transcribed to DNA using a specific enzyme.
Additional short fragments of DNA that are complementary to specific parts of the transcribed viral DNA are added to the sample. If the virus is present in a sample, these fragments attach themselves to target sections of the viral DNA. Some of the added genetic fragments are used for building DNA strands during amplification, while the others are used for adding marker labels to the strands, which are then used to detect the virus.
The mixture is then placed in an RTPCR machine. The machine cycles through temperatures that heat and cools the mixture to trigger specific chemical reactions that create new, identical copies of the target sections of the viral DNA. The cycle is repeated over and over to continue copying the target sections of the viral DNA. Each cycle doubles the previous number.
A standard real time RTPCR set-up usually goes through 35 cycles, which means that, by the end of the process, around 35 billion new copies of the sections of the viral DNA are created from each strand of the virus present in the sample.
As new copies of the viral DNA sections are built, the marker labels attach to the DNA strands and then release a fluorescent dye, which is measured by the machines computer and presented in real time on the screen. The computer tracks the amount of fluorescence in the sample after each cycle. When a certain level of fluorescence is surpassed, this confirms that the virus is present.
Scientists also monitor how many cycles it takes to reach this level in order to estimate the severity of the infection: the fewer the cycles, the more severe the viral infection is. It is the most reliable test for COVID-19. The time duration for the testing process is around four to eight hours, but results may take up to a day to arrive because of the time taken for other process.
2. Antigen Testing
These tests are designed to detect a specific protein in the virus that elicits the bodys immune response. In the case of COVID-19, it is the spike protein present on the surface of the coronavirus that facilitates its entry into the human cell.
For this test, a nasal swab is collected, which is then immersed in a solution that deactivates the virus. A few drops of this solution are then put on a test strip. This has to be done within an hour of the immersion of the swab in the solution.
The test strips contain artificial antibodies designed to bind to coronavirus proteins. If a person is infected with coronavirus, the test lines will appear on the paper strips within 15 minutes.
Since antigen testing does not involve any amplification process, swab samples may lack enough antigen material to be detectable. This may give false negative results. For this reason, if a person tests negative through antigen testing, they still need to get an RT-PCR test done for confirmation. If a person tests positive, however, a confirmation RT-PCR is not required.
The new antigen tests that have been approved for COVID-19 diagnosis in India give results in 30 minutes. Its main advantage is that it reduces the burden of relying on just RT-PCR tests to identify COVID-19 patients.
3. TrueNat
It is used in Semi quantitative detection of Beta Coronavirus (Sarbeco), RNA in human oropharyngeal and nasopharyngeal swab specimen. Betacoronaviruses (â-CoVs or Beta-CoVs) are one of four genera (Alpha-, Beta-, Gamma-, and Delta-) of coronaviruses. They are enveloped, positive-sense, single-stranded RNA viruses that infect human and mammals.
From the betacoronaviruses, greatest clinical importance concerning humans are OC43 and HKU1 (which can cause the common cold) of lineage A, SARS-CoV and SARS-CoV-2 (which causes the disease COVID-19) of lineage B and MERS-CoV of lineage C.
The test is recommended for use as a first line screening test for COVID-19. TrueNat machine can detect an enzyme (called RdRp) found in the RNA of the virus. RNA dependent RNA polymerase (RdRp) is one of the most versatile enzymes of RNA viruses that is indispensable for replicating the genome as well as for carrying out transcription.
Samples testing positive by Truenat Beta CoVshould be confirmed using confirmatory tests for SARS CoV 2.
4. Antibody Tests (Serological Testing)
Antibody testing (or serological testing) is an examination of proteins in the bloodstream, to find out whether an individual has been infected with COVID-19. An infected person will have specific antibodies to pathogens they have been exposed to. The immune system produces antibodies as part of a larger process to defend itself from an infection.
That said, antibody tests may not be able to show whether the virus is currently infecting the body. Unlike a nasal or throat swab test, which looks for genetic signatures of the virus in the body, an antibody test looks for traces of the bodys response to the virus.
Antibodies are abundant in the blood, so a sample of blood is collected by either a finger prick or a blood sample drawn with a needle. Two specific antibody types are sought out in an antibody test:
i) IgM antibodies against SARS-CoV-2, which develops early on in an infection.
ii) IgG antibodies against SARS-CoV-2, which are mostly found after someone has recovered from the infection.
This test have high margin of error as of now. It is mainly used for research and surveillance purposes.
Summary:
* Hanjabam Rajanikanta Sharma wrote this article for The Sangai Express
The writer is a Former intern at IIT Delhi,
and did his BS-MS (Bio Major) from Indian Institute of Science Education and Research (IISER), Mohali.
He can be reached at rajhanjabam(AT)yahoo(DOT)com
This article was webcasted on August 01 2020 .
* Comments posted by users in this discussion thread and other parts of this site are opinions of the individuals posting them (whose user ID is displayed alongside) and not the views of e-pao.net. We strongly recommend that users exercise responsibility, sensitivity and caution over language while writing your opinions which will be seen and read by other users. Please read a complete Guideline on using comments on this website.