Introduction
2005 Nobel Prize in Chemistry has been awarded to Frenchman Yves Chauvin and Americans Richard Schrock and Robert Grubbs for their contributions to a reaction process called metathesis1. A brief account of the research activities of the three Nobel Laureates who shared the US $1.3 millions is presented in this article.
YVES CHAUVIN
- Born: 1930
- Place of Birth: France
- Citizenship: France
- Residence: France
- Affiliation: Institut Francais du Petrole
- Address: 1 & 2 Avenue de Bois-Preau, 92852 Rueil-Malmaison,France
ROBERT H. GRUBBS
- Born: February 27, 1942
- Place of Birth: Calvert City, Kentucky, USA
- Citizenship: USA
- Residence: USA
- Affiliation: California Institute of Technology, Pasadena, CA, USA
RICHARD R. SCHROCK
- Born: January 4, 1945
- Place of Birth: Berne, IN, USA
- Citizenship: USA
- Residence: USA
- Affiliation: Massachusetts Institute of Technology, Cambridge, MA, USA
Introduction
Synthetic organic chemists have presently at their disposal a plethora of synthetic methods for the elaboration of simple organic compounds to many complex and sophisticated compounds. The enormous diversity of organic molecules that has been explored by synthetic chemists has given us new pharmaceuticals, agrochemicals, materials etc. that we cannot live without. In recognition of the contributions towards these reaction approaches, five of the Nobel Prizes in chemistry has been awarded so far to the following synthetic organic chemists:
(i) the 1912 Prize to V. Grignard and P. Sabatier, for discovery of the Grignard reagents in forming carbon-carbon bonds for the construction of molecular frameworks and the use of metals in catalytic hydrogenation of unsaturated compounds;
(ii) the 1950 Prize to Otto Diels and Kurt Alder for the discovery of the Diels-Alder reaction which involves the formation of new carbon-carbon bonds from dienes and dienophiles;
(ii) the 1979 Prize to H.C. Brown and G. Wittig for their contributions to the chemistry of the carbon-carbon bond (hydroboration of unsaturated carbon-carbon bonds and the chemistry thereof) and its formation (the Wittig reaction);
(iv) the 2001 Prize to W.S. Knowles and R. Noyori (reduction of the double bond) and to K.B. Sharpless (oxidation of the double bond) using asymmetric catalysis, and this year
(v) the 2005 Nobel Prize in Chemistry has been awarded to Dr. Yves Chauvin at the Institut Français du Pétrole, Rueil-Malmaison, France and Professor Robert H. Grubbs, California Institute of Technology, Pasadena, California, USA and Professor Richard R. Schrock, Massachusetts Institute of Technology, Cambridge, Massachusetts, USA for their contributions to a reaction process called metathesis. A brief account of the historical perspectives of the metathesis with special emphasis on the pioneering works of the three Nobel laureates is given in the first part of this article. The second part of the article devotes to the various practical applications of metathesis in pharmaceuticals, hydrocarbons and polymer industries.
Metathesis Reactions
The word metathesis is derived from the Greek meta (change) and tithemi (place). As a grammatical term it means the transposition of sounds or letters in a word. In chemistry it refers to the interchange of atoms between two molecules. Organic substances contain the element carbon. Carbon atoms can form long chains and rings, bind other elements such as hydrogen and oxygen, form double bonds, etc. All life on Earth is based on these carbon compounds, but they can also be produced artificially through organic synthesis. Metathesis is also a kind of organic synthesis where many carbon compounds from the very simple and small molecular weight ones to very large, complex polymers, macromolecules and even natural products are synthesized. The substrates, which convert into products, are in general olefins i.e. carbon compounds containing double bonds and based on this the process is also known as "olefin metathesis". In metathesis reactions, double bonds are broken and made between carbon atoms in ways that cause atom groups to change places. This happens with the assistance of special catalyst molecules. Metathesis can be compared to a dance in which the couples change partners. The metathesis of olefins is the reaction in which the molecules of these hydrocarbons are formally fragmented at their double bonds and new olefin molecules result by recombination of fragments originating from different molecules as shown in scheme-1:

Scheme - 1
If A=B=C=H, and D=CH3, then comes the organic molecule called propene and two molecules of propene undergo olefin metathesis with the help of a catalyst, producing two new alkenes, butene and ethene (ethylene).
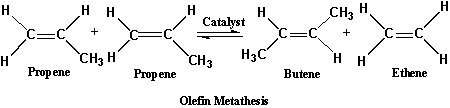
Scheme - 2
In this metathesis reaction one of the propene molecules exchanges its CH2 group for the CH3CH group in the other propene molecule. The result is butene and ethene. A catalyst, which is not consumed in the reaction, is required for the reaction to occur. The ring opening polymerization of cyclic olefins, which is also known as metathesis polymerization, is the reaction by which such molecules are cleaved at the double bond producing unsaturated linear polymers.
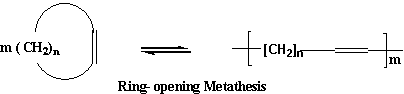
Scheme - 3
In addition to the above two types, newer interesting forms of metathesis were developed. Scheme-4 shows the application of metathesis in ring formation reactions, which is known as ring-closing metathesis.
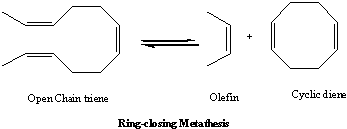
Scheme - 4
The cross metathesis is another form of metathesis where a group of new olefins can be prepared from two or more olefins as shown here:
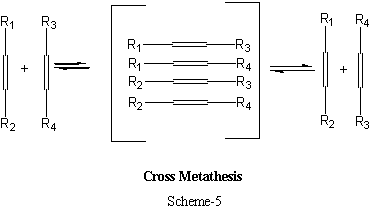
Cross-metathesis is one of the widely used synthetic methods of polymers and an extended form of this type of metathesis is shown in Scheme-6.
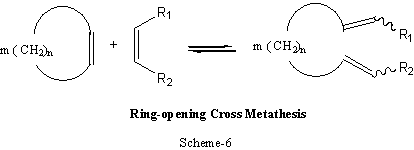
The chemical reaction schemes 2-6, are generally reversible and, with the right catalyst system, equilibrium can be attained in a matter of seconds, even with substrate /catalyst ratios of 104; a truly remarkable reaction.
The Chauvin mechanism
In 1970, Chauvin and Herisson2 suggested a chain mechanism for metathesis and ring opening polymerization in the homogeneous phase, through carbonic intermediates. Chauvin also presented an entirely new mechanism for how the metal compound functions as a catalyst in the reaction. His new experimental results tallied with this new mechanism and could not be explained by any previously proposed mechanism.
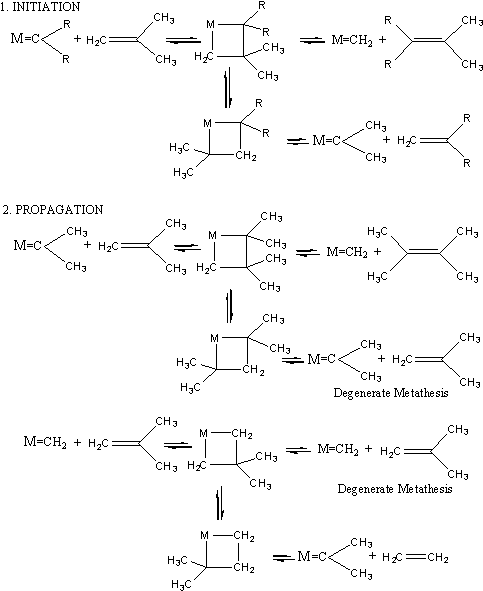 Scheme-7 (Chauvin s mechanism, proposed in 1971, for the catalyzed olefin metathesis involving metal alkylidene and metallacyclobutane intermediates).
Scheme-7 (Chauvin s mechanism, proposed in 1971, for the catalyzed olefin metathesis involving metal alkylidene and metallacyclobutane intermediates).
Scheme- 7 shows the mechanism. In the first stage of the reaction metal methylene combines with one alkene to form a ring of four atoms. The ring consists of the metal atom and three carbon atoms bound to one another with single bonds. In the next step two of the single bonds are broken and a new alkene (ethene) and a new metal alkylidene is obtained. The new metal-alkylidene contains one of the two carbenes of the starting olefin and it can re-enter into a catalytic cycle of the same type as the first one. In fact, depending on the orientation of the coordinated olefin, the new catalytic cycle can give two different metallacyclobutenes, one leading to the symmetrical olefin and the other one leading the starting olefin. This latter cycle is said to be degenerate olefin metathesis. Thus, the catalytic cycles alternatively involve both metal-alkylidene species resulting from the combination of the metal with each of the two carbenes of the starting olefin.
Chauvin and Hérisson not only suggested the metallacyclobutane mechanism, but also published several experiments to confirm it. For instance, the reaction of a mixture of cyclopentene and 2-pentene led to C-9, C-10 and C-11 dienes in the ratio 1 2 1. Also, the reaction of a mixture of cyclooctene and 2-pentene led almost exclusively to the C-13 product. The latter reaction, but not the first one, was compatible with Calderon s mechanism. In 1973, Chauvin published other results showing that the WCl6 + MeLi mixture catalyzes the formation of propene by reaction of 2-butene, which was proposed to proceed via methylation of tungsten, followed by the  -elimination in the tungsten-carbon bond of W-CH3 to form W CH2 (H), then metathesis.3 The carbonic chain mechanism suggested by Chauvin was supported by the new experimental results obtained by many authors, however, the most strong support for the mechanism was from experimental investigations by Robert H. Grubbs4 and Richard R. Schrock.5 -elimination in the tungsten-carbon bond of W-CH3 to form W CH2 (H), then metathesis.3 The carbonic chain mechanism suggested by Chauvin was supported by the new experimental results obtained by many authors, however, the most strong support for the mechanism was from experimental investigations by Robert H. Grubbs4 and Richard R. Schrock.5
Grubbs and Schrock,s Contributions in Developing catalyst molecules
Catalyst systems for the olefin metathesis generally contain a transition metal compound. These are sometimes effective by themselves but often require the presence of a second compound (co catalyst), and sometimes a third (promoter). The system most commonly used are based on the chlorides, oxides, or other easily accessible compounds of Mo, Ru, W, Re, Os, Ir, Ti, V, Cr, Co, Nb, Rh, or Ta compounds. Typical co catalysts are EtAlCl2, R3AlCl2, R3Al and R4Sn(R=Ph, Me, Et, Bu), while promoters often contain oxygen, e.g. O2, EtOH, PhOH. More and more chemists now started to realise that metathesis could assume great importance for organic synthesis if reliable and effective catalysts could be found. Earlier, undefined catalysts were used, sensitive to air and moisture and also relatively short-lived. The requirement was stable and well-defined catalysts with reactivity that could be adjusted depending on the purpose. In addition they had to be selective - only react with double bonds and leave other parts of the molecules intact. Chauvin's results showed how efficient metal alkylidene catalysts could be constructed. The problem was that none of the known well defined metal alkylidenes acted as catalysts in olefin metathesis. A number of chemists made major contributions to the development of metathesis catalysts and their applications; but the crucial progress in this area was made by Robert H. Grubbs and Richard R. Schrock.
Richard Schrock started research on new alkylidene complexes in the early 1970s. For Schrock, molybdenum and tungsten soon appeared to be the most suitable metals. Some catalysts were produced with those metals, but there was still uncertainty as to what groups would bind to the metal to give stable yet active alkylidene complexes. A breakthrough came in 1990 when Schrock and co-workers reported4-5 the construction of a group very active, well-defined molybdenum catalyst (fig. 1).
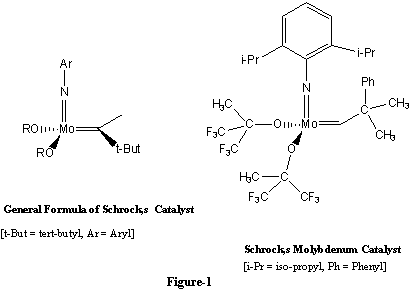
With this discovery chemists began to realize that olefin metathesis could be used for general purposes in organic synthesis. Metathesis gained increasing attention among researchers active in synthetic chemistry. It turned out that metathesis can replace a number of traditional synthesis methods. At the same time it permits entirely new approaches to the synthesis of organic molecules.
Yet another breakthrough in the development of metathesis catalysts came in 1992 when Robert Grubbs and his co-workers published6-7 their discovery of a catalyst with the metal ruthenium. It was stable in air and demonstrated higher selectivity but lower reactivity than Richard R. Schrock's catalysts. The new catalyst also had the ability to initiate metathesis in the presence of alcohols, water and carboxyl acids (compare with Fig. 2). Grubbs afterwards improved his catalysts and in Fig. 2, one of his effective metathesis catalysts that are easy to synthesize are shown.
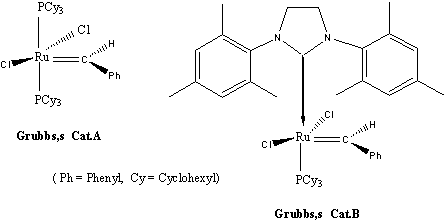
Figure 2
Grubbs' catalysts have become the first well-defined catalysts for general metathesis applications in ordinary laboratories. Catalyst 2 in fig. 2 is generally named Grubbs' catalyst and has become a standard with which all new catalysts are compared. The general applicability of Grubbs' catalyst has given rise to future prospects of the possibilities of organic synthesis. Grubbs bases his catalyst design on detailed mechanical studies. He has continued development of ruthenium-based metathesis catalysts into yet more powerful tools for synthesis, including that of polymers with special properties.
Applications of Metathesis
The metathesis reaction discovered less than twenty years ago has become soon afterwards a large and independent research area. It is applied previously in the synthesis of olefins and functionalized olefins, of monomers, in the formation of plastics and elastomers, and in petrochemistry. After the discovery of its extraordinary potential for the synthesis of molecular targets used as therapeutic agents, the entire organic community and pharmaceutical companies use it. Nowadays, metathesis is at the forefront of green chemistry owing to shorter synthetic paths and providing more facile access to therapeutic agents under increasingly more environmentally friendly catalytic conditions. This gives us novel possibilities to exploit the enormous diversity of organic molecules and contributes to a "greener" chemistry. Metathesis is significant to green chemistry because:
(1) Reactions are more efficient (fewer steps, fewer resources, less waste)
(2) Reactions are easier to use (stable under ambient conditions)
(3) Environmentally benign (non-hazardous solvents, less hazardous waste). For example, Grubbs used a green approach, from RuCl3 in water to the sophisticated chiral version of the second generation ruthenium benzylidene catalyst, to provide the organic and polymer communities with what are now the most environmentally friendly metathesis catalysts, because of their stability to air and even to some extent to aqueous media and their high compatibility with most organic functionalities.
Conclusion
The pioneering works of Yves Chauvin has brought a new shape in metathesis with a successful mechanistic approach. The mechanism of Chauvin was supported unambiguously by Schrock,s experimental works. Schrock has contributed to the chemical community the first stable metal-methylene and -alkylidene complexes. Many organic chemists started exploring the applications of Schrock s chiral catalysts to asymmetric synthesis. Similarly, Grubbs provides the organic and polymer communities the air and water stable second generation ruthenium benzylidene catalysts with what are now the most environmentally friendly metathesis catalysts. Schrock-type and Grubbs-type metathesis catalysts are also complementary in terms of efficiency, compatibility of functional groups and stability.
Together, the laureates' contributions have brought a major significance in the chemicals industry, opening up new opportunities for synthesizing molecules that will streamline the development and industrial production of pharmaceuticals, plastics and other materials.
References
1. Anon., The 2005 Nobel Prize in Chemistry. Nobel Prize press release, http://nobelprize.org/chemistry/laureates/2005/press.html
2. Y. Chauvin and J.-L. Hérisson, Makromol. Chem., 1971, 141, 161-176.
3. Chauvin, Y. C. R. Seances Acad. Sci., Ser. C, 1973, 276, 169; (b) Chauvin, Y., Commereuc, D. and D. Cruypelinck, Makromol. Chem., 1976, 177, 2637-2647.
4. (a) Schrock, R. R., Murdzek, J.S., Barzan, G.C., Robbins, J., DiMare, M. and O`Regan, M. J. Am. Chem. Soc. 1990, 112, 3875-3886; (b) Teng, X., Cefalo, D. Schrock, R. R. and Hoveyda, A. H. J. Am. Chem. Soc., 2002, 124, 10779-10784.
22. Nguyen, S. T., Johson, L. K., Grubbs, R. H. and Ziller, J. W. J. Am. Chem. Soc.,
1992, 114, 3974-3975.
5. (a) Schrock, R. R. and Parshall, G. W. Chem. Rev., 1976, 76, 243-268 (b) Schrock, R. R. Acc. Chem. Res., 1979, 12, 98-104; (c) Schrock, R. R., Science, 1983, 219, 13-18; (d) Schrock, R. R. J. Am. Chem. Soc., 1974, 96, 6796-6797; (e) Schrock, R. R., Rocklage, S. M. , Wengrovius, J. H., Rupprecht, G. and Feldmann, J. J. Mol. Catal., 1980, 8, 73-83; (f) J. H. Wengrovius, J., Schrock, R. R., Churchill, M. R. , Missert, J. R. and Youngs, W. J. J. Am. Chem. Soc., 1980, 102, 4515-4516. (g) Wood, C.D., McLain, S.J. and Schrock, R. R., J. Am. Chem. Soc. 1979, 101, 3210-3222. (h) Murdzek, J.S. and Schrock, R. R., Organometallics 1987, 6, 1373-1374.
6. Nguyen, S. T., Johson, L. K. , Grubbs, R. H. and Ziller, J. W. J. Am. Chem. Soc., 1992, 114, 3974.
7. (a) Handbook of Metathesis, ed. Grubbs, R. H. Wiley-VCH, Weinheim, 2003, Vols.
1-3:Catalyst Development (vol. 1); Applications in Organic Synthesis (vol. 2); Applications in Polymer Synthesis (vol. 3); (b) For a major account of Ru metathesis catalysts, see: Nguyen, S. T. and Trnka, T. M. in Handbook of Metathesis, ed. Grubbs, R. H. Wiley-VCH, Weinheim, 2003, vol. 1, ch. 1.6; (c) Trnka, T. M. and Grubbs, R. H. Acc. Chem. Res., 2001, 34, 18-29 (this article also contains many references to reviews); (d) For mechanistic aspects of Ru metathesis catalysts, see Sanford, M. S. and Love, J. in Handbook of Metathesis, ed. Grubbs, R. H. Wiley-VCH, Weinheim, 2003, vol. 1, ch. 1.9; (e) Grubbs, R. H. Tetrahedron, 2004, 60, 7117-7140 (this review article also contains the references to more than 400 Grubbs articles); (f) Grubbs, R. H. and Chang, S. Tetrahedron, 1998, 54, 4413-4450.
|












 Scheme-7 (Chauvin s mechanism, proposed in 1971, for the catalyzed olefin metathesis involving metal alkylidene and metallacyclobutane intermediates).
Scheme-7 (Chauvin s mechanism, proposed in 1971, for the catalyzed olefin metathesis involving metal alkylidene and metallacyclobutane intermediates).